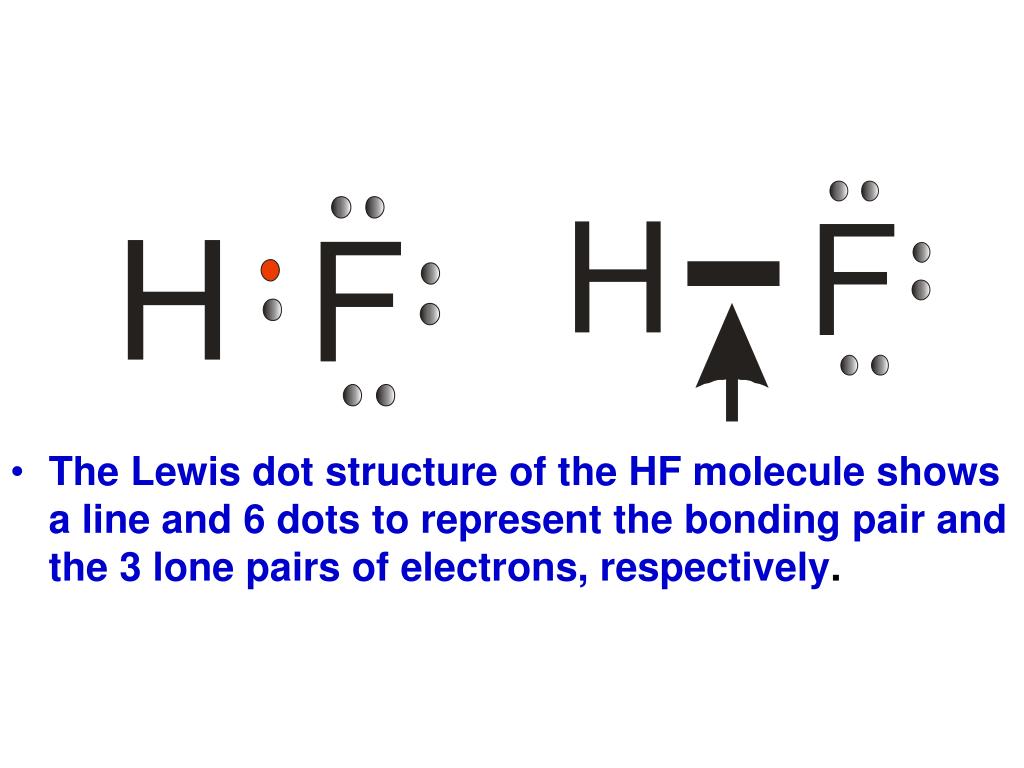
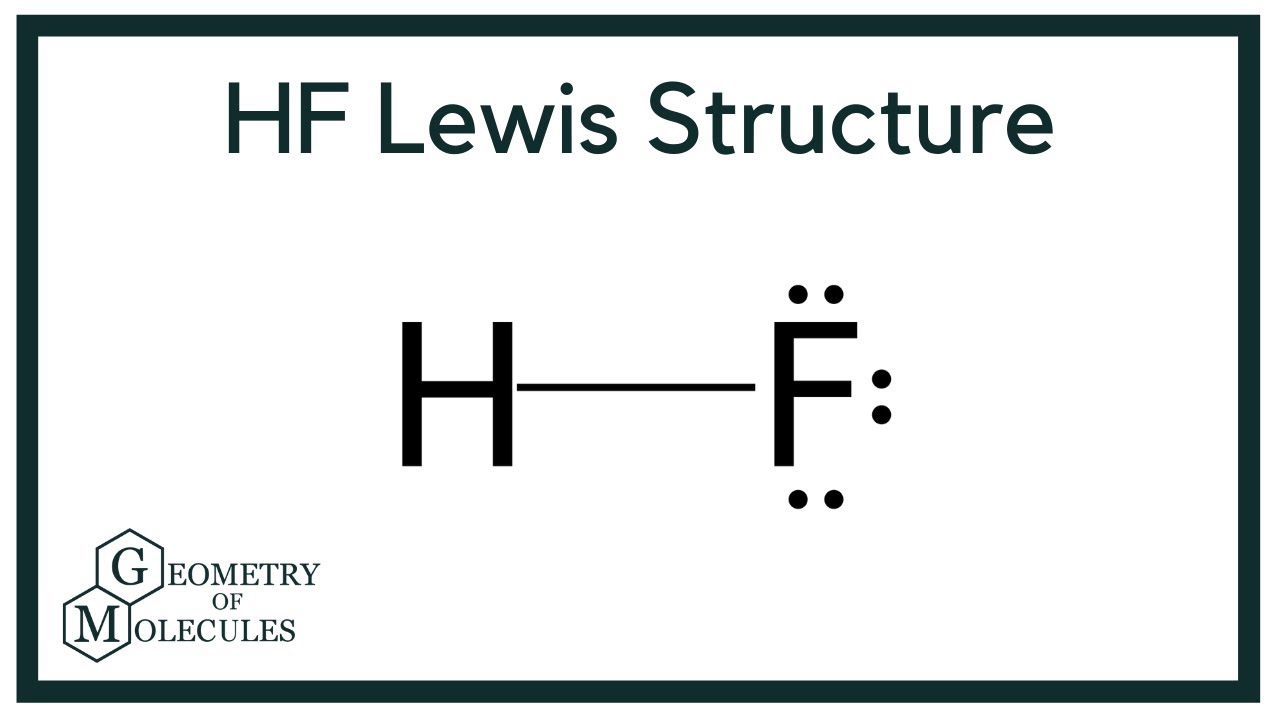
Draw The Lewis Structure Of Hf
Draw The Lewis Structure Of Hf - Here, the given molecule is hf (hydrogen fluorine). Web we can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using lewis dot symbols: C is the central atom since it makes the most. In the case of hf, we can represent the valence electrons of hydrogen and fluorine using dots around their respective atomic symbols. Web draw the lewis structure for i) co ii) xef4 iii) pcl3;
Web steps of drawing hf lewis structure step 1: The central atom is fluorine, which is bordered on two terminals with hydrogen atoms ( in. Web we can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using lewis dot symbols: Web steps for writing lewis structures. Hf is very similar to hf and hcl. With the lewis structure for hf remember that hydrogen only needs 2. Select the center atom (h is always outside).
Hydrogen fluoride Lewis structure Covalent bond Chemical bond, others
Web 0:00 / 2:03 hf lewis structure (hydrogen fluoride) geometry of molecules 2.71k subscribers subscribe 4 share 717 views 1 year ago lewis structure hello everyone! Draw the lewis structure for p 4. Web draw the lewis structure for i) co ii) xef4 iii) pcl3; In hf, the fluorine atom is more electronegative than hydrogen.
How to draw HF Lewis Structure? Science Education and Tutorials
Select the center atom (h is always outside). Draw the lewis structure for p 4. Use lewis electron dot diagrams to illustrate the covalent bond formation in hbr. Count the total number of valence electrons in the molecule. While selecting the atom, you have to put the least. Draw the lewis structure for sof_4. Draw.
HF Molecular Geometry Science Education and Tutorials
Web 6 steps to draw the lewis structure of hf step #1: Web we can illustrate the formation of a water molecule from two hydrogen atoms and an oxygen atom using lewis dot symbols: Draw the lewis structure for p 4. Count the total number of valence electrons in the molecule. Calculate the sum of.
Identify a bond between H and F in HF molecule Brainly.in
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Calculate the sum of the valence electrons in the molecule. The structure on the right is the lewis electron structure, or lewis structure, for h 2 o. Select the center atom (h is always outside). Draw the lewis structure.
Lewis dot structure of HF Hydrofluoric acid lewis dot structure
Hydrogen has 1 valence electron and fluorine (in group 7 with f and cl) has 7 valence electrons. Web i quickly take you through how to draw the lewis structure of hf (hydrogen fluoride). Assign formal charges to atoms in the structure. Calculate the total number of valence electrons. For the hf structure use the.
PPT Lewis Dot Structures of Covalent Compounds PowerPoint
Determine the central atom in the molecule. Web here’s how you can easily draw the hf lewis structure step by step: I also go over the shape and bond angle. Web some structures don't obey the octet rule, but explain why. Key points to consider when drawing the hf electron dot structure. Reference the “how.
So far, we’ve used 8 of the HF Lewis structure’s total 8 outermost
We have to first count the total number of valence electrons inside a single hydrogen fluoride molecule. Web steps for writing lewis structures. Here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. Web we can illustrate the formation of a water molecule from.
Draw the Lewis Structure of HF (hydrogen fluoride) YouTube
Hydrogen has 1 valence electron and fluorine (in group 7 with f and cl) has 7 valence electrons. With the lewis structure for hf remember that hydrogen only needs 2. Web 67k views 10 years ago. Draw the lewis structure for sof_4. With two bonding pairs and two lone pairs, the oxygen atom has now.
HF Lewis Structure (Hydrogen Fluoride) YouTube
In hf, the fluorine atom is more electronegative than hydrogen and. Construct a skeleton structure for the molecule. Here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. Hydrogen has 1 valence electron and fluorine (in group 7 with f and cl) has 7.
HF Lewis Structure, Molecular Geometry, Hybridization, and Polarity
Draw the lewis structure for p 4. Construct a skeleton structure for the molecule. Hydrogen has 1 valence electron and fluorine (in group 7 with f and cl) has 7 valence electrons. Draw the lewis structure of hydronium (h3o+) and then determine its electrons domain and. The central atom is fluorine, which is bordered on.
Draw The Lewis Structure Of Hf Web drawing the lewis structure for hf. Web how to draw the lewis structure of hf hydrogen fluoride?lewis structure: Reference the “how to draw a. Here, we will be using the determined total number of valence electrons per atom and drawing them in the proper places. Draw lewis dot structures for ch4 ch 4, nh3 nh 3, hf hf, of2 of 2, f2 f 2, o2 o 2, n2 n.
Hydrogen Has 1 Valence Electron And Fluorine (In Group 7 With F And Cl) Has 7 Valence Electrons.
Key points to consider when drawing the hf electron dot structure. C is the central atom since it makes the most. While selecting the atom, you have to put the least. We have to first count the total number of valence electrons inside a single hydrogen fluoride molecule.
Draw The Lewis Structure For Sof_4.
Hf is very similar to hf and hcl. Reference the “how to draw a. Web the two atoms can share their unpaired electrons to make a covalent bond: Note that the h atom has a full valence shell with two electrons, while the f atom has a complete octet of electrons.
Web How To Draw The Lewis Structure Of Hf Hydrogen Fluoride?Lewis Structure:
Web some structures don't obey the octet rule, but explain why. Web science chemistry chemistry questions and answers draw the lewis structure for hf this problem has been solved! Web 6 steps to draw the lewis structure of hf step #1: For the hf structure use the periodic table to find the total number of.
In This Video, We Are.
Use lewis electron dot diagrams to illustrate the covalent bond formation in hbr. I also go over the shape and bond angle. Draw the lewis structure for hf draw the lewis structure for hf here’s the best way to solve it. Web how to draw hf lewis structure?