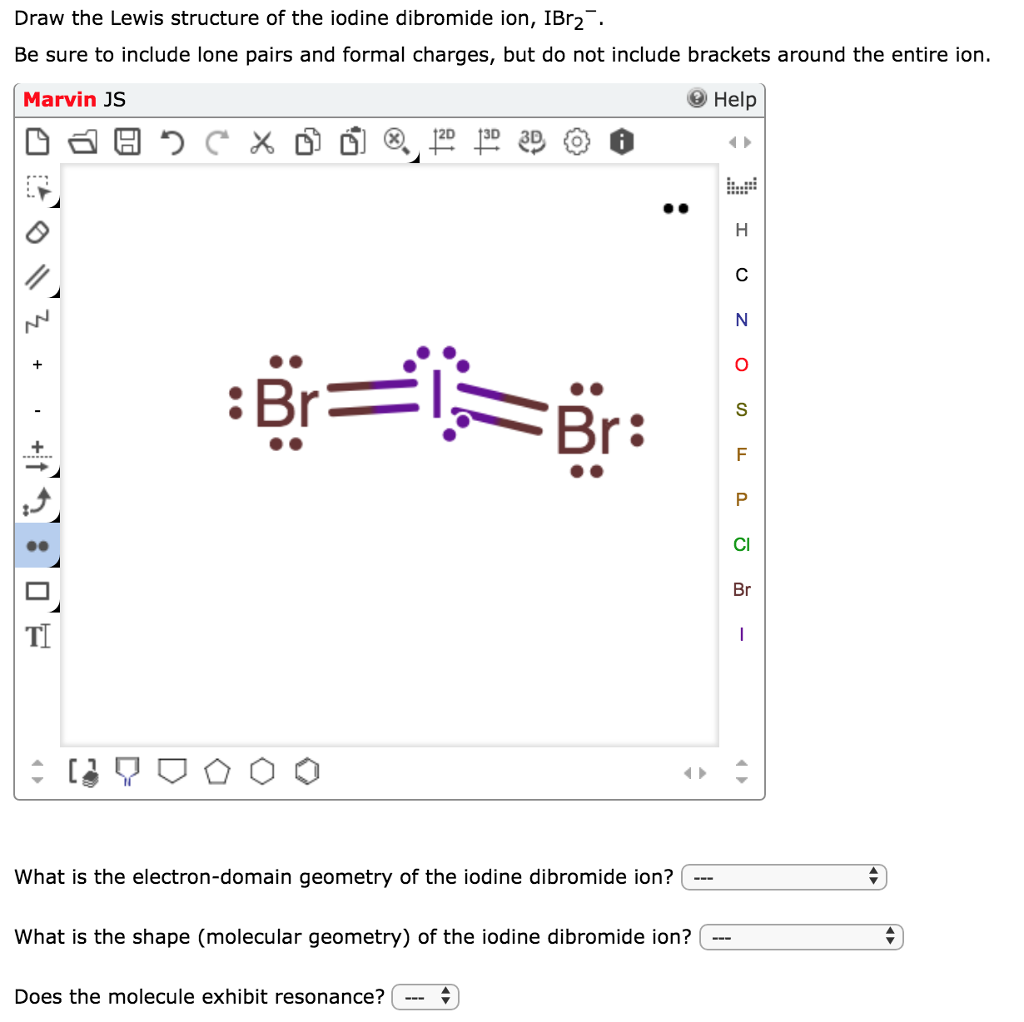
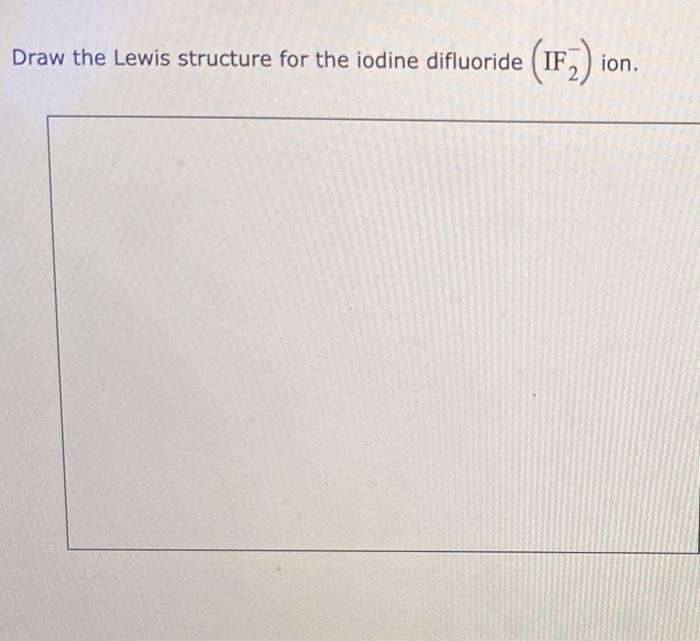
Draw The Lewis Structure For The Iodine Difluoride Ion
Draw The Lewis Structure For The Iodine Difluoride Ion - First, we must connect all atoms with a single bond, then we place the remaining electrons as. This problem has been solved! Web draw the lewis structure for the iodine difluoride (if,) ion. Web concept explainers question please complete the lewis structure transcribed image text: Web write lewis symbols for neutral atoms and ions.
See an example of a molecule that violates the octet rule (xef₂) and learn how to draw its lewis diagram in this video. The structure of hydrogen has to be drawn. Web chemistry chemistry questions and answers draw the lewis structure for the iodine difluoride (if2−)ion. Draw lewis structures depicting the bonding in simple molecules. Web the need for the number of electrons lost being equal to the number of electrons gained explains why ionic compounds have the ratio of cations to anions that they do. C expert solution trending now this is a popular solution! That is s e o f two.
How To Draw The Lewis Dot Structure For I Iodide Ion
Draw lewis structures depicting the bonding in simple molecules. That is s e o f two. This is required by the law of conservation of matter as well. Web draw the lewis structure for the iodine difluoride (if,) ion. There will be four billion electrons plus my chin, which has watson group five. Which statements.
(Get Answer) Draw The Lewis Structure For The Iodine Difluoride (IF 7
Let's put that in the center and then fluorines can go on the outside. Web to draw a lewis structure we first need to find the total number of electrons in a lewis structure, which is equal to the sum of valence electrons of every atom in the molecule. A lewis diagram shows how the.
Lewis Dot Diagram Iodine
This problem has been solved! This problem has been solved! Hydrogen has an elected configuration. This problem has been solved! When constructing a lewis diagram, keep in mind the octet rule, which refers to the tendency. Web drawing lewis structures for molecules with one central atom: Fluorine also has 7, but we have two fluorines;.
Solved Draw the Lewis structure for the iodine difluoride
Let's put that in the center and then fluorines can go on the outside. Web drawing lewis structures for molecules with one central atom: (note, all the atoms are bonded to the central i atom.) steric number octet rule exceptions choose at least one answer. Web draw the lewis electron structure of the molecule or.
Lewis Structure For Iodine
Lewis diagram of xenon difluoride (xef₂) in some molecules, the central atom exceeds the octet rule (is surrounded by more than eight electrons). Ć ö | с [ х s ? Draw the lewis structure for the iodine difluoride ( if, ion. When constructing a lewis diagram, keep in mind the octet rule, which refers.
The gallery for > Xef2 Vsepr
You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Determine the electron group arrangement around the central atom that minimizes repulsions. There will be four billion.
Lewis Structure For Iodine
You're starting from the first part. In section 4.7, we demonstrated that ions are formed by losing electrons to make cations, or by gaining electrons to form anions. There will be four billion electrons plus my chin, which has watson group five. You'll get a detailed solution from a subject matter expert that helps you.
Iodine Lewis Dot Diagram
Web concept explainers question please complete the lewis structure transcribed image text: Thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Let's put that in the center and then fluorines can go on the outside. Shared pairs of electrons are drawn as lines between atoms,.
Iodine Electron Configuration (I) with Orbital Diagram
Ć ö | с [ х s ? Web to draw a lewis structure we first need to find the total number of electrons in a lewis structure, which is equal to the sum of valence electrons of every atom in the molecule. This problem has been solved! Lewis diagram of xenon difluoride (xef₂) in.
Draw the Lewis structure for the iodine difluoride IF… SolvedLib
This electron dot diagram helps us understand the atomic structure , electron configuration , and chemical bonding of iodine. You're starting from the first part. Draw lewis structures depicting the bonding in simple molecules. Draw the lewis structure for the iodine difluoride (if 7 ) in ion. So it's five bills are transferred, five minus.
Draw The Lewis Structure For The Iodine Difluoride Ion Lewis diagram of xenon difluoride (xef₂) in some molecules, the central atom exceeds the octet rule (is surrounded by more than eight electrons). Web concept explainers question please complete the lewis structure transcribed image text: See an example of a molecule that violates the octet rule (xef₂) and learn how to draw its lewis diagram in this video. Web concept explainers question transcribed image text: Assign an ax m e n designation;
Synthesis Of Calcium Chloride From Elements.
The astute reader may have noticed something: Draw lewis structures depicting the bonding in simple molecules. Lewis diagram of xenon difluoride (xef₂) in some molecules, the central atom exceeds the octet rule (is surrounded by more than eight electrons). Web draw the lewis structure for the iodine difluoride (if,) ion.
This Is Required By The Law Of Conservation Of Matter As Well.
Olo c ar | expert solution trending now this is a popular solution! And then we have an extra valence electron from that negative sign for a total of 22 valence electrons. In this question, we have a problem. So it's five bills are transferred, five minus the number of electrons squared.
Ć Ö | С [ Х S ?
Draw the lewis structure for the iodine difluoride (if2−)ion. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw the lewis structure for the iodine difluoride ( if, ion. Let's put that in the center and then fluorines can go on the outside.
This Problem Has Been Solved!
Web concept explainers question please complete the lewis structure transcribed image text: Hydrogen has an elected configuration. Web write lewis symbols for neutral atoms and ions. C expert solution trending now this is a popular solution!