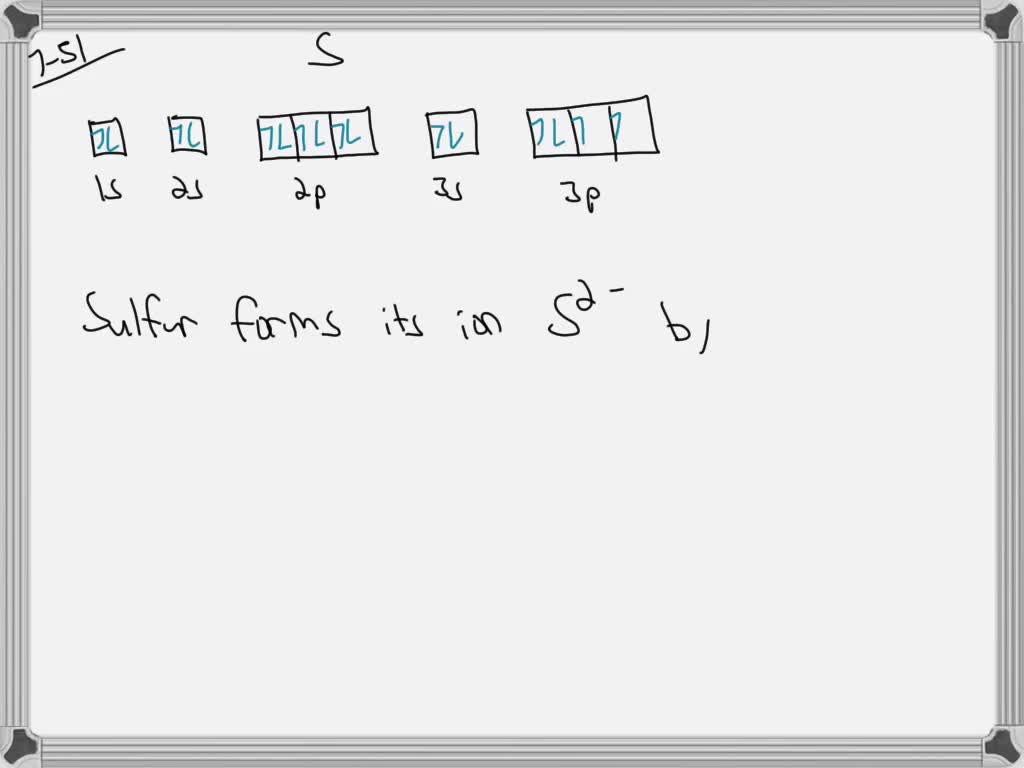Draw The Electron Dot Formula For The Element Sulfur
Draw The Electron Dot Formula For The Element Sulfur - Include all of the valence electrons. Web continuing with sulfur, we observe that in (a) the sulfur atom shares one bonding pair and has three lone pairs and has a total of six valence electrons. Include all of the valence electrons. You can add the valence electrons by clicking on the or button and clicking the atom. Include all of the valence electrons.
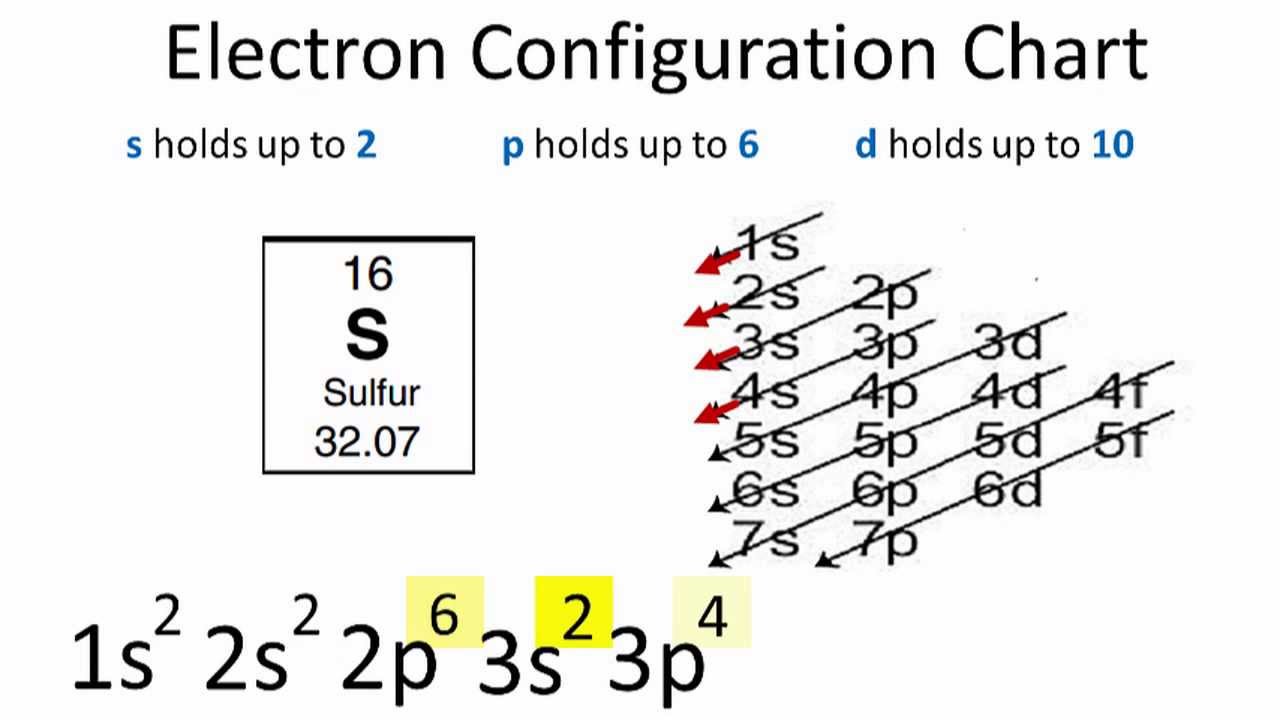
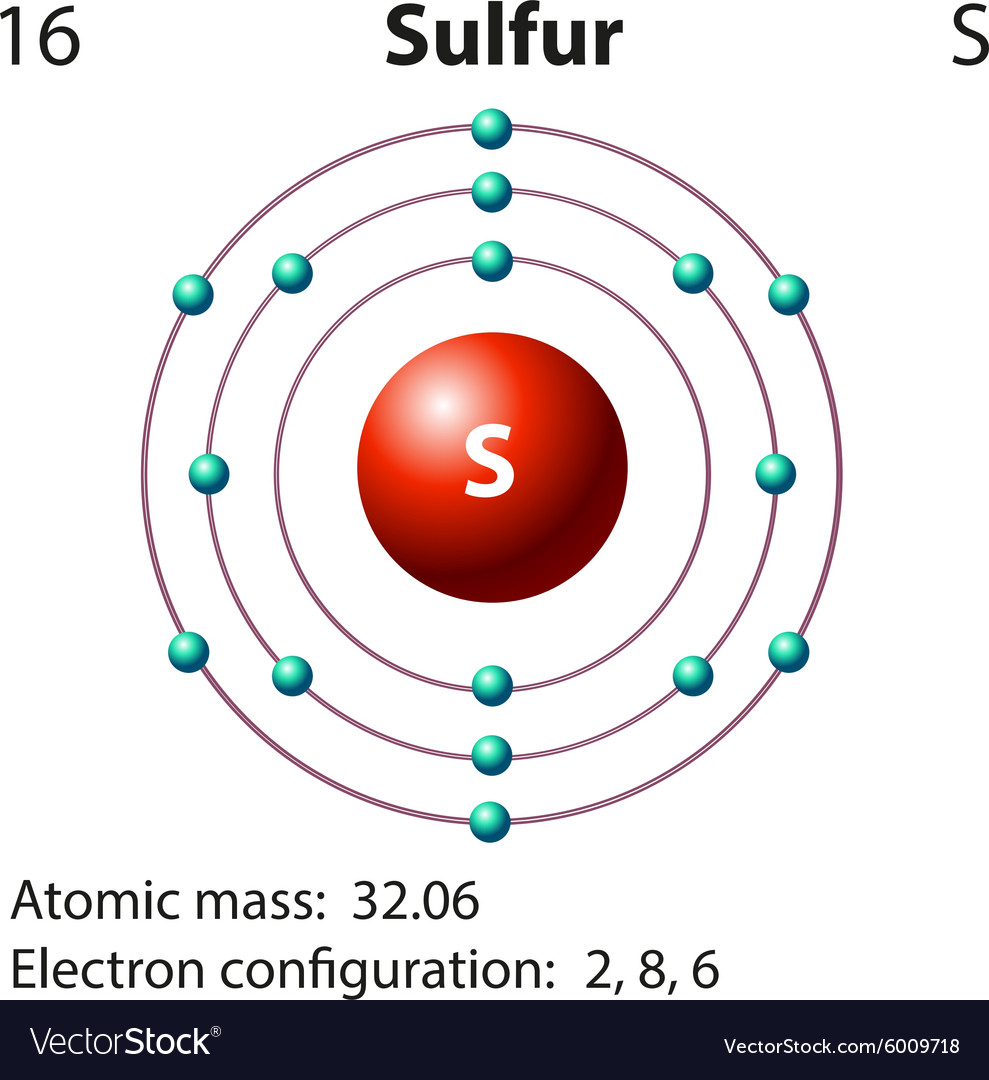
Sulfur , which is located in group 6a, has 6 valence electrons. For sulfur there are six valence electrons in 3s and 3p shell. Lewis dot structure of sulfur atom sulfur ion lewis dot structure We'll put the s, the least electronegative, at the center. Web a lewis electron dot diagram (or electron dot diagram, or a lewis diagram, or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. The lewis electron dot diagram would look like the following: Web sulfur represents properties such as dryness, heat, and masculinity.
Atom Diagrams Electron Configurations of the Elements
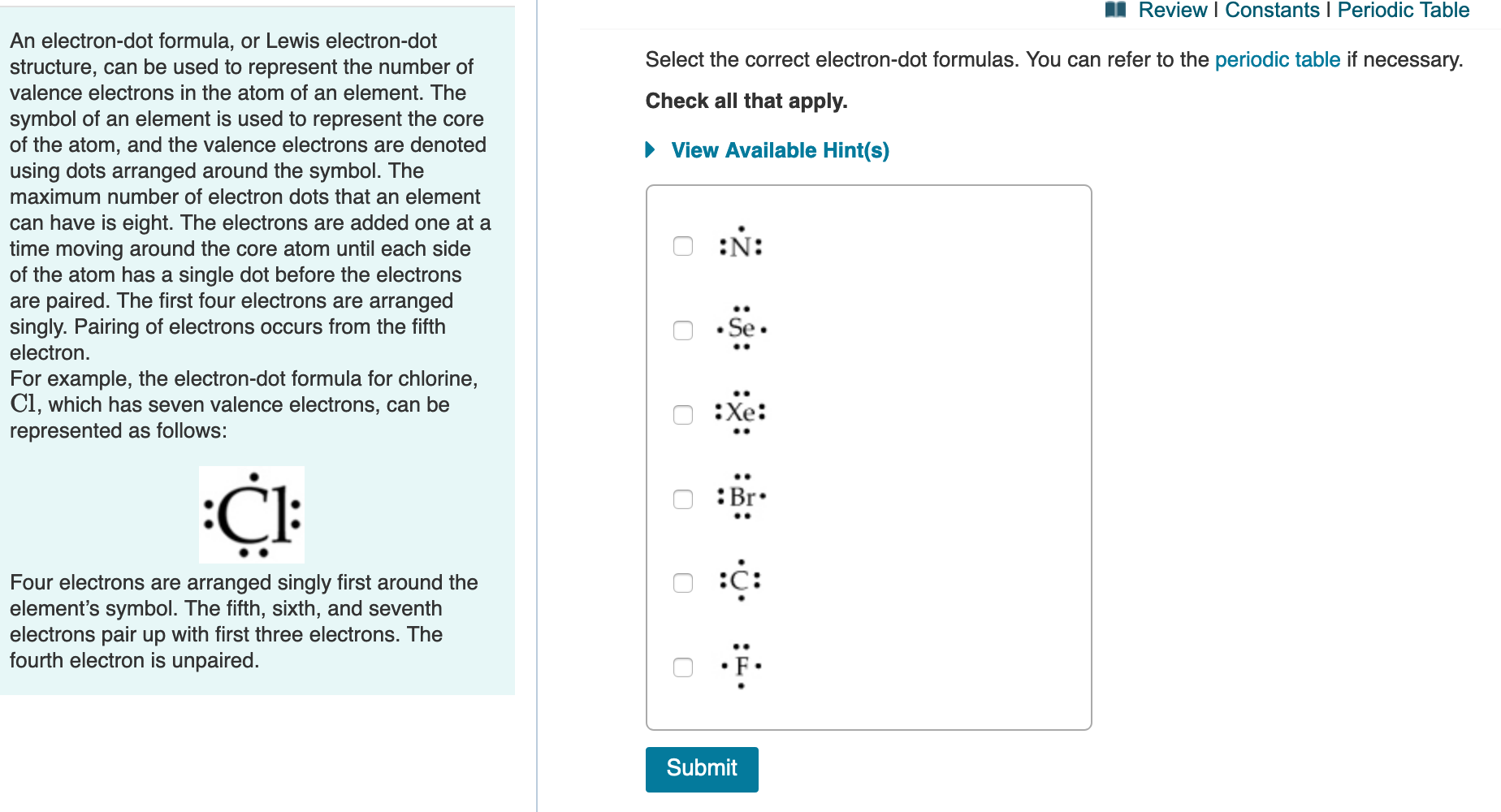
It has a very simple chemical formula, h 2 o. Include all of the valence electrons. Therefore, chlorine has 7 valence electrons. The number of dots equals the number of valence electrons in the atom. The electrons are drawn in a clockwise manner starting on the right. You'll get a detailed solution from a subject.
Sulfur S (Element 16) of Periodic Table Elements FlashCards
Web a lewis electron dot diagram (or electron dot diagram, or a lewis diagram, or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. In terms of the tria prima, sulfur was seen as the middling element connecting salt (high) and mercury.
Sulfur Electron Configuration (S) with Orbital Diagram
Web the core electrons would be 1 s2 2 s2 2 p6 while the valence electrons would be in the third shell (or where n = 3). Web a lewis electron dot symbol (or electron dot diagram or a lewis diagram or a lewis structure) is a representation of the valence electrons of an atom.
Sulfur Definition, Facts, Symbol, Allotropes, Properties, Uses
The most famous allotrope that exists from these elements is a diamond, which is an allotrope of carbon. Figure 7.9 shows the lewis symbols for the elements of the third period of the periodic table. The next electron is drawn underneath the symbol, and then on the left side. To eliminate sodium, an element essential.
Solved Draw the electrondot formula for the element
Include all of the valence electrons. Lewis symbols illustrating the number of valence electrons for each element in the third period of the periodic table. Sulfur , which is located in group 6a, has 6 valence electrons. Lots of things dissolve easily in water. · s ····↙·↘···· in the case of sulfur , which is.
Sulfur Atom Science Notes and Projects
The next electron is drawn underneath the symbol, and then on the left side. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Therefore, chlorine has 7 valence electrons. We'll put the s, the least electronegative, at the center. This problem has been solved! Web only five elements.
SOLVEDThe orbital notation of sulfur is shown in Figure 7.15. Explain
The most famous allotrope that exists from these elements is a diamond, which is an allotrope of carbon. The number of dots equals the number of valence electrons in the atom. In terms of human body, it represented the soul. 100% (1 rating) transcribed image text: · s ····↙·↘···· in the case of sulfur ,.
Draw the electrondot formula for the element sulphur YouTube
Include all of the valence electrons. The lewis electron dot diagram would look like the following: Water is remarkable in terms of all the things it can do. Include all of the valence electrons. The element sulfur (s) has six valence electrons in its outer shell. Tin, phosphorus, sulfur, oxygen, and carbon. Since fluorine is.
Diagram representation of the element sulfur Vector Image
Web a lewis electron dot diagram (or electron dot diagram, or a lewis diagram, or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. For sulfur there are six valence electrons in 3s and 3p shell. You can add the valence electrons.
draw electron dot structure of the molecule of sulphur which is made up
In terms of human body, it represented the soul. Web only five elements are known to have allotropes: Web to draw the electron dot structure of sulfur atom write the atomic symbol of sulfur that is “s”. Web chemicals in the soil. We'll put the fluorines around it, all four of them. To eliminate sodium,.
Draw The Electron Dot Formula For The Element Sulfur Web only five elements are known to have allotropes: 100% (1 rating) transcribed image text: Web to draw the electron dot structure of sulfur atom write the atomic symbol of sulfur that is “s”. We'll put the fluorines around it, all four of them. Water is remarkable in terms of all the things it can do.
Web Let's Do The Sf4 Lewis Structure.
In alchemy, it could also represent evaporation, expansion, and dissolution. You can add the valence electrons by clicking on the or button and clicking the atom. You can add the valence electrons by clicking on the or button and clicking the atom. Web water is an amazing molecule.
Web Draw A Valid Electron Dot Structure For Each Of The Given Elements.
Web to draw the electron dot structure of sulfur atom write the atomic symbol of sulfur that is “s”. Web continuing with sulfur, we observe that in (a) the sulfur atom shares one bonding pair and has three lone pairs and has a total of six valence electrons. Web the core electrons would be 1 s2 2 s2 2 p6 while the valence electrons would be in the third shell (or where n = 3). Some types of rock can even completely dissolve in water ( figure below)!
Web Chemicals In The Soil.
Web a lewis electron dot symbol (or electron dot diagram or a lewis diagram or a lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. You can add the valence electrons by clicking on the or button and clicking the atom. The formal charge on the sulfur atom is therefore 6 −(6 + 2 2) = −1.5 −(4 + 4 2) = −1 in (c), nitrogen has a formal charge of −2. In terms of the tria prima, sulfur was seen as the middling element connecting salt (high) and mercury (low).
Web Only Five Elements Are Known To Have Allotropes:
For a total of 34 valence electrons. Tin, phosphorus, sulfur, oxygen, and carbon. Lewis dot structure of sulfur atom sulfur ion lewis dot structure To eliminate sodium, an element essential to human health but harmful to plants, sulfur needs a little help from calcium, another element.

:max_bytes(150000):strip_icc()/sulfuratom-58b602563df78cdcd83d5a9d.jpg)